Reducing Risk: Rethinking healthcare design and operations to improve infection control
By Mike Apple, Jim Atkinson & Susan McDevitt
Infection control in hospitals and healthcare facilities has historically been focused on reducing the spread of pathogenic microorganisms from patient to patient, or from staff to patient, to reduce the impact of hospital-acquired or healthcare-associated infections. Over the last decade, there has been an increasing focus on preventing the spread of infectious diseases such as the seasonal flu within hospitals and healthcare facilities; however, this has taken place primarily through operational measures rather than facility design strategies.
The risks to patients, families and healthcare workers during the COVID-19 pandemic has resulted in a significant rethinking of facility design strategies in conjunction with operational practices to limit the transmission of the virus and further reduce other HAIs. This rethinking has resulted in the best practices that follow.
Enhancing disease screening and building entry options
The most effective infection control strategies may indeed be clean hands and universal masking. At each entry and throughout the facility, culture, communication and signage should encourage mask use with effective filtration and proper fit. Universal screening may be more appropriate for inpatients, labor and delivery patients and procedural patients. A separate entry should be provided for patients known to be COVID-19 positive.
If a hospital does choose to perform some type of universal screening at entry, it should occur in a well-ventilated space or even outdoors if possible. The amount of space needed for screening depends on the number of persons entering and the time required for screening. Appropriate, socially distanced queuing space may be required. Since the screening process has a significant impact on safety and visitor and staff satisfaction, the facility should regularly update processes based on fluctuations in volume, community infection rates and updates in clinical evidence.
Improving infection control and efficiency in areas frequented by patients
The design for inpatient and emergency departments moving forward requires flexibility; however, that flexibility must be scalable. The specific needs for the isolation of infectious patients depends on the pathogen, transmission methodologies and the quantity of infectious patients. The facility response should be layered to allow for multiple scenarios and pathways for well and infectious patients.
Institutions are creating screening and holding spaces within or adjacent to the emergency department for suspect cases to be confirmed prior to moving the patient to the appropriate care area. These spaces can be designed to provide access from outside and from ambulance entries to minimize the risk of suspected cases freely moving about the facility. Anteroom entries can be provided for appropriate airflow control and donning and doffing of PPE to ensure staff safety and containment of infectious material within this dedicated space.
PPE use and facility design
The almost universal use of personal protective equipment such as face masks, shields, respirators, gloves, gowns and shoe coverings as appropriate for the care setting adds a time and efficiency burden to the staff that may be impacted by facility design.
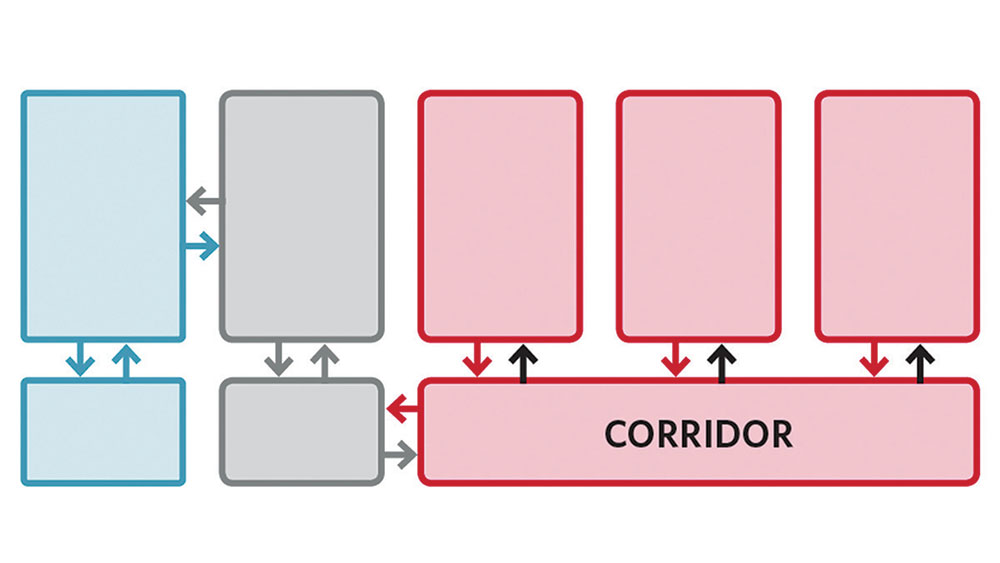
Traditionally, single-patient isolation rooms have been planned for most types of patient care units. These negative pressure isolation rooms should include an anteroom for hand-washing, donning and doffing of PPE, and as a buffer to maintain proper airflows. While this approach used to be the only mechanism to isolate a patient on a unit, it now represents a template that can be activated at larger scales.
If several infectious patients are being treated simultaneously for the same pathogen, a pod of rooms can be converted into a dedicated isolation zone. This zone would be defined by cross-corridor doors and have dedicated ventilation. A common anteroom would be employed for entering and exiting the pod, while the corridor accessing the patient rooms would now be the buffer zone to properly maintain airflow. This approach extends the life of PPE and maximizes direct patient care time because staff may only need to change their gloves between each patient and their outer PPE when contaminated.
If a large cohort of patients with the same infection must be isolated, the pod approach can flex up to the scale of an entire patient unit. Careful planning for HVAC separation and redundancy, clean and dirty flows and off-stage access should be integrated at all scales of isolation.
Rethinking the control and elimination of infectious aerosols
Flexibility is key for rooms that have a high turnover of patients that may be potentially infected, such as exam areas in EDs or in ambulatory care facilities. Infectious pathogens such as COVID-19 with potential contact, droplet and aerosol transmission create a challenge for room utilization due to the need to decontaminate the surfaces and air between room use.
Surface decontamination can be accomplished by wiping down with sanitizing agents or by installing UV lighting systems that inactivate infectious organisms. Large droplets tend to fall out of the airstream rapidly and can be inactivated by the same methods.
For small aerosol droplets the issue is more complicated. Infectious aerosols can remain suspended for long periods and can also be potentially transmitted through recirculated heating, ventilating and air conditioning systems. Typical air changes in these rooms are not adequate for the removal of these agents. For example, CDC guidance suggests a room with 2-4 air changes per hour will take 2-3 hours to remove 99.9% of airborne contaminants, and to achieve a 10-minute room turn-around time it could take up to 50 ACH. With conventional HVAC systems, these levels of removal would be cost prohibitive both in construction and energy cost.
The quantity of aerosolized pathogens in room air and in air moving into the corridor is reduced by both staff and patients wearing PPE, and by achieving directional airflow (rebalancing supply and return air). Aerosols are removed from the room using a HEPA-filtered air cleaner sized for rapid cleaning with off- and low-speed options for exam acoustic needs and high speed for air cleaning between patients. An advantage of this approach is that it does not require additional conditioning of air and only uses energy when on unless outside exhaust is required.
If built-in air cleaners are not practical, portable units on wheels are available that could be brought into a room between patient use to filter the air to CDC recommendations within 10 minutes. Again, energy is only used when the air cleaner is in use. A strategy of low-air changes from the HVAC system, HEPA filtration of return air, and air cleaning by room recirculation through a HEPA-filtered unit allow higher room turnover rates with low-operating cost and energy use.
It will be interesting to study the impact of precautions for COVID-19 on other more routine HAIs through an analysis of data on infections before and after the pandemic. This might inform the healthcare industry what measures and design strategies should be kept in place for future pandemics or the general goal of reducing the normal pattern of HAIs.
Author: Mike Apple, Jim Atkinson & Susan McDevitt
Mike Apple, RA, ACHA, LSSGB, is a health planner at HDR. Jim Atkinson, AIA, ACHA, EDAC, LEED AP, is healthcare planning director at HDR. Susan McDevitt, MSN, ARNP, EDAC, LSSGB, is clinical planning principal at HDR.
Editor’s Note: Jon Crane, FAIA, LEED AP, director of translational health sciences at HDR, contributed to this article.
Posted May 27, 2021
More Articles:
- Coverings 2024
Apr 22, 2024 – Apr 25, 2024 - Hospital, Outpatient Facilities & Medical Office Buildings Summit
Apr 25, 2024 – Apr 25, 2024 - CxA Workshop & Exam
Apr 29, 2024 – Apr 30, 2024 - EMP Seminar & Exam at CxEnergy 2024
Apr 29, 2024 – Apr 30, 2024 - CxEnergy
Apr 29, 2024 – May 2, 2024 - PHCC West 2024
Apr 29, 2024 – May 2, 2024 - Lean in Design Forum 2024
May 1, 2024 – May 2, 2024